Clinical Trial Protocol Training Template For Nurse Coordinators
Clinical Trial Protocol Training Template For Nurse Coordinators - New hire and onboarding resources, core. Welcome to global health trials' tools and templates library. Personnel to assess site readiness to begin a clinical trial. A protocol is a document that describes the background, rationale, objectives, design, methodology, statistical considerations, and organization of a trial. Please note that this page has been updated for 2015 following a quality check and review of the templates, and many new ones. A group of senior research nurses and research coordinators are working to develop standard operating procedures for clinical trial management and conduct according to the good clinical. Protocol guidance and template for use in a. The clinical trial coordinator orientation and competency manual adopted by the ctsu embraces immediate guidance and support for staff involved in clinical trials. This generally includes a tour of the facilities, meeting most staff assigned to the study, and protocol training. It may be useful as an example for centres and organisations who are new to clinical trials. The clinical trial coordinator orientation and competency manual adopted by the ctsu embraces immediate guidance and support for staff involved in clinical trials. This is an example of a costing template used by trials at mrc ctu. This generally includes a tour of the facilities, meeting most staff assigned to the study, and protocol training. Personnel to assess site readiness to begin a clinical trial. New hire and onboarding resources, core. Clinical trials conducted after regulatory submission of a dossier, but prior to the medicine's approval and launch. Phase 2 or 3 clinical trials that require. A group of senior research nurses and research coordinators are working to develop standard operating procedures for clinical trial management and conduct according to the good clinical. The clinical trial coordinator orientation and competency manual adopted by the ctsu embraces immediate guidance and support for staff involved in clinical trials. Click on each box to be taken. Personnel to assess site readiness to begin a clinical trial. To ensure comprehensive training for clinical research coordinators, covering key aspects of their role and responsibilities in conducting successful clinical trials. Click on each box to be taken. Protocol guidance and template for use in a. A group of senior research nurses and research coordinators are working to develop standard. This is an example of a costing template used by trials at mrc ctu. A protocol is a document that describes the background, rationale, objectives, design, methodology, statistical considerations, and organization of a trial. Phase 2 or 3 clinical trials that require. To ensure comprehensive training for clinical research coordinators, covering key aspects of their role and responsibilities in conducting. Ucsf has created a comprehensive checklist of training steps for clinical researchers, which breaks training activities apart into three categories: To ensure comprehensive training for clinical research coordinators, covering key aspects of their role and responsibilities in conducting successful clinical trials. A protocol is a document that describes the background, rationale, objectives, design, methodology, statistical considerations, and organization of a. This is an example of a costing template used by trials at mrc ctu. Ucsf has created a comprehensive checklist of training steps for clinical researchers, which breaks training activities apart into three categories: Protocol guidance and template for use in a. New hire and onboarding resources, core. The clinical trial coordinator orientation and competency manual adopted by the ctsu. Personnel to assess site readiness to begin a clinical trial. This generally includes a tour of the facilities, meeting most staff assigned to the study, and protocol training. The clinical trial coordinator orientation and competency manual adopted by the ctsu embraces immediate guidance and support for staff involved in clinical trials. A protocol is a document that describes the background,. To ensure comprehensive training for clinical research coordinators, covering key aspects of their role and responsibilities in conducting successful clinical trials. Added senior research coordinator to data quality monitoring table. Protocol guidance and template for use in a. This generally includes a tour of the facilities, meeting most staff assigned to the study, and protocol training. The clinical trial coordinator. Welcome to global health trials' tools and templates library. It may be useful as an example for centres and organisations who are new to clinical trials. Ucsf has created a comprehensive checklist of training steps for clinical researchers, which breaks training activities apart into three categories: Nih applicants can use a template with instructional and sample text to help write. A protocol is a document that describes the background, rationale, objectives, design, methodology, statistical considerations, and organization of a trial. Nih applicants can use a template with instructional and sample text to help write clinical protocols for the following types of research: Protocol guidance and template for use in a. The clinical trial coordinator orientation and competency manual adopted by. Protocol guidance and template for use in a. A group of senior research nurses and research coordinators are working to develop standard operating procedures for clinical trial management and conduct according to the good clinical. The clinical trial coordinator orientation and competency manual adopted by the ctsu embraces immediate guidance and support for staff involved in clinical trials. Please note. The onboarding learning plan templates include: A protocol is a document that describes the background, rationale, objectives, design, methodology, statistical considerations, and organization of a trial. This is an example of a costing template used by trials at mrc ctu. This generally includes a tour of the facilities, meeting most staff assigned to the study, and protocol training. Ucsf has. Added senior research coordinator to data quality monitoring table. Personnel to assess site readiness to begin a clinical trial. A group of senior research nurses and research coordinators are working to develop standard operating procedures for clinical trial management and conduct according to the good clinical. To ensure comprehensive training for clinical research coordinators, covering key aspects of their role and responsibilities in conducting successful clinical trials. Click on each box to be taken. A protocol is a document that describes the background, rationale, objectives, design, methodology, statistical considerations, and organization of a trial. These trials may supplement earlier trials, complete earlier trials, or may. Please note that this page has been updated for 2015 following a quality check and review of the templates, and many new ones. Welcome to global health trials' tools and templates library. The onboarding learning plan templates include: This is an example of a costing template used by trials at mrc ctu. Changed sample size from 180 enrolled to 210 enrolled, and included updated power analysis for this sample. The clinical trial coordinator orientation and competency manual adopted by the ctsu embraces immediate guidance and support for staff involved in clinical trials. Protocol guidance and template for use in a. This generally includes a tour of the facilities, meeting most staff assigned to the study, and protocol training. It may be useful as an example for centres and organisations who are new to clinical trials.Clinical Trial Protocol Template Word
Clinical Study Protocol (CSP) Template Clinical Study Templates
Clinical Trial Protocol Template Word
Clinical Study Protocol PowerPoint and Google Slides Template PPT Slides
Fillable Online clinicaltrials.govProvidedDocs66CIRB Clinical Trial
Clinical Study Protocol Template
Free Clinical Trial Templates Smartsheet
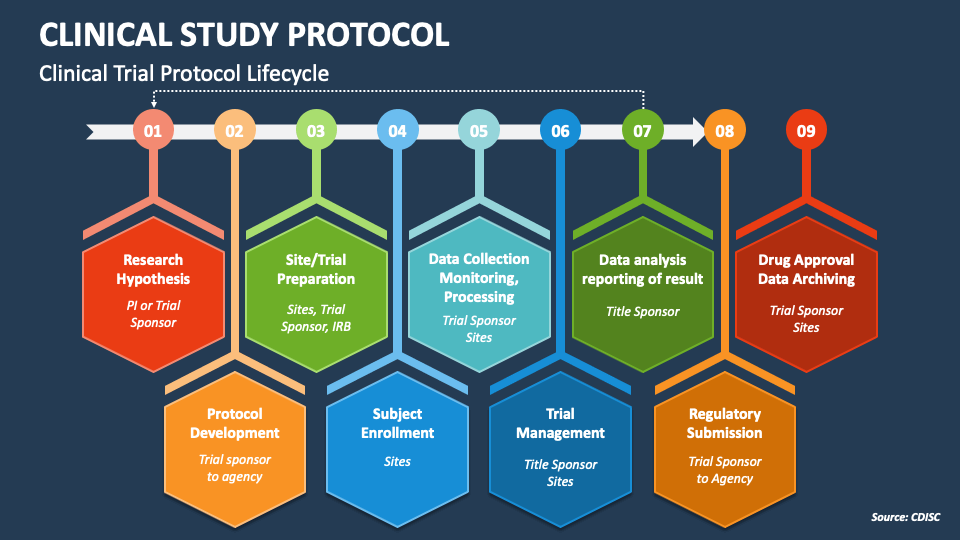
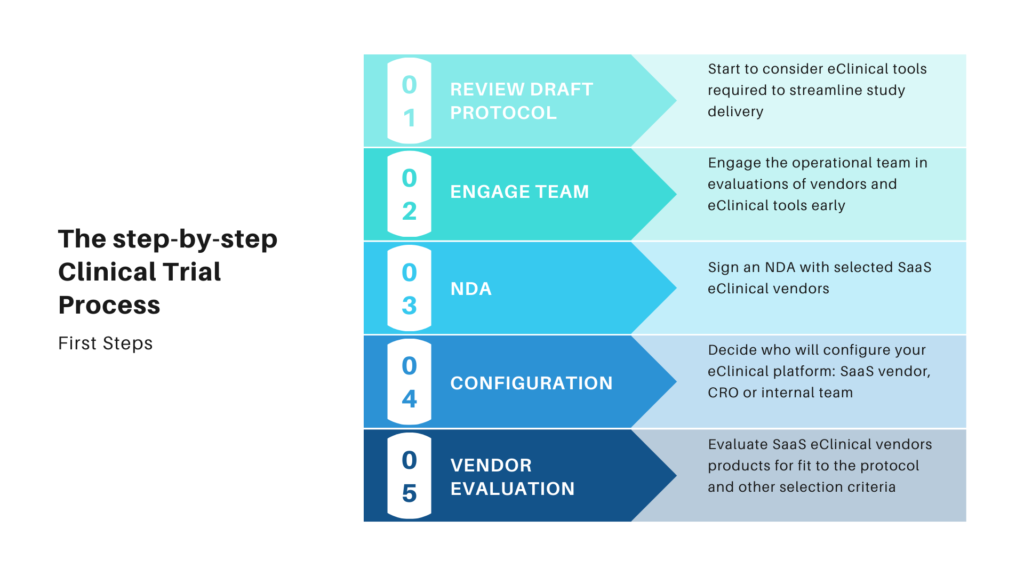
The Clincial Trial Process Stepbystep approach
Clinical Trial Protocol Template Health Interventions Doc Template
(PDF) Clinical Trial Protocol Template€¦ · GCP Good Clinical Practice
Nih Applicants Can Use A Template With Instructional And Sample Text To Help Write Clinical Protocols For The Following Types Of Research:
Phase 2 Or 3 Clinical Trials That Require.
New Hire And Onboarding Resources, Core.
Click On Each Box To Be.
Related Post: